19
th
Annual ENETS Conference| 10 – 11 March 2022
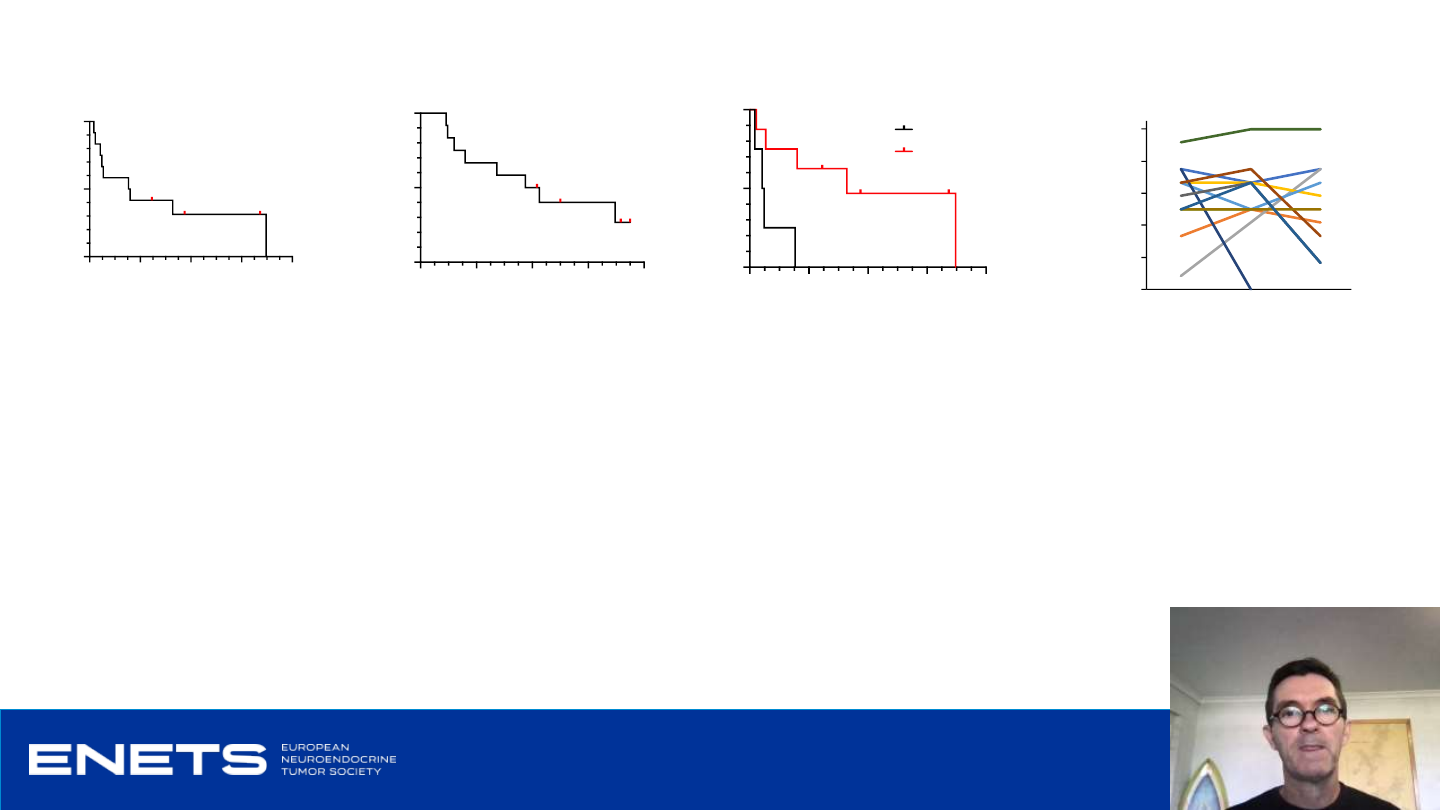
Secondary endpoints: PFS , overall survival and QOL
Median PFS 9.4 months
• Range 1.0 to 41.8 months
0
12
24
36
48
0
50
100
Months
O
ve
ra
ll
S
u
rv
iv
al
(
%
)
Data 1
Median OS 22.5 months
• Range 5.5 to 42.3 months (4 subjects alive)
0
12
24
36
48
0
50
100
Survival proportions: Survival of Data 2
Months
P
FS
(%
)
0
12
24
36
48
0
50
100
Survival proportions: PFS by Histology
Months
P
FS
(%
)
Pancreas
Intestine
Primary Histology
• Median PFS 2.7 mon vs 19.7 mon
• Median OS 11.8 mon vs 25.5 mon
0
20
40
60
80
100
Baseline Month 1 Month 3
Global Health Status
PV-10 elicited no safety concerns and multiple doses were safely delivered.
Encouraging evidence of both local and systemic disease and symptom control was seen in a heavily pre -
treated population.
PV-10 may be an option for patients who have failed standard therapy.
Combining PV-10 with systemic checkpoint inhibitors may enhance the proposed immune mechanism.
Non-clinical data in other poorly immunogenic tumour types suggests PV-10 may enhance activity of cytotoxic
therapies, such as PRRT.
Conclusion